EST. 2014
Leveraging Polymerization Equilibrium to Make (and Unmake) Elastomeric New Materials


The Complexity in Ring Strain Energies
Ring strain energies can vary greatly as a function of cycloalkanes (bars), cycloalkenes (dashes) and the number of carbons that constitute the ring (n)
As (n) increases, entropy also plays an important role. The carbon bonds within a ring gain rotational entropy when opened into the linear polymeric state. This is assists in reducing the entropic penalty associated with molecular aggregation of monomers during all polymerizations.
Variable Temperature ROMP of Cyclopentenes
controlling molar mass and dispersity
Cyclopentene (CP) has a low ring strain and gains very little rotational entropy during ROMP. Therefore, these systems have been notoriously difficult to obtain targeted molar mass, low dispersity, and high conversion, simultaneously. We developed a method that leverages the thermodynamic principles above and the unique catalyst behavior of Grubbs 1st generation catalyst (G1) at varying temperatures. Through high temperatures initiation and low temperature propagation, which we call VT-ROMP, high molecular weight and low dispersity polypentenamers were produced at targeted molar mass and high conversions for the first time.


This discovery is a universal approach to gaining high levels of macromolecular control over functionalized cyclopentene derivatives with suitable ring strain energy.


Unmaking Polypentenamers through Ring Closing Metathesis Depolymerization (RCMD)
architectural metamorphosis
The same thermodynamic principles that make CP derivatives challenging for ROMP also make them highly suited to undergo depolymerization through ring closing metathesis (RCM). We leveraged this principle to create singular bottlebrush systems with polypentenamer backbones capable of unzipping through RCM; thereby producing linear grafts with CP end groups that could be coupled to thiols and produce star polymers. This unique progression through several architectural derivatives starting from a singular type of polypentenamer opens the door to a variety of stimuli-responsive materials.
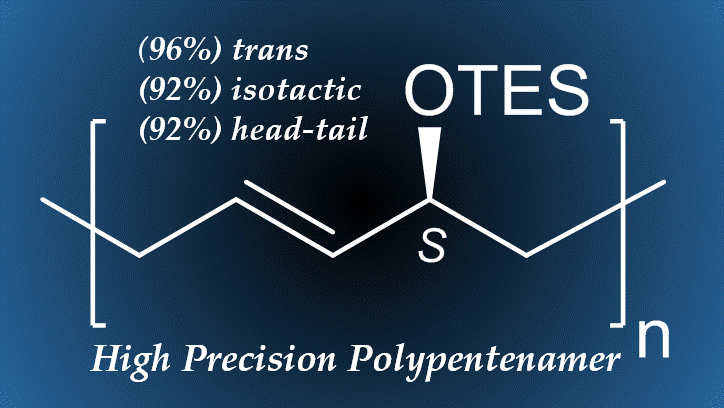
Higher Precision Polymers
controlling stereo- and regio-chemistries
Gaining control over the stereochemistry and regioregularity of polymers is a historically difficult yet sought after task. A methodology, using bulky allylic substituents on cyclopentene monomers has recently been developed that allows for control over the tacticity and regioregularity of the polymer. Hence isotactic branches at every five carbons along a polypentenamer rubber are produced. Such materials are predicted to have complex material and thermal properties which are being investigated.
To gain isotacticity, we must use allylic substituted CP monomers. The combination of low ring strain and steric effects of these substituents presents very intriguing sensitivity to the thermodynamics of ROMP. Pictured here is how the equilibrium monomer concentration, [M]eq, varies as a function of temperature for a series of different bulky substituents on CP. The table inset shows that both the enthalpy and entropy are highly sensitized to these steric effects.